
Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

You know the right answer?
Write the electron configuration for . (Express your answer as a series of orbitals, in order of inc...
Questions

Spanish, 31.08.2019 16:30

History, 31.08.2019 16:30




Mathematics, 31.08.2019 16:30

Mathematics, 31.08.2019 16:30

Mathematics, 31.08.2019 16:30


Biology, 31.08.2019 16:30

Social Studies, 31.08.2019 16:30

Physics, 31.08.2019 16:30




Mathematics, 31.08.2019 16:30

Social Studies, 31.08.2019 16:30

History, 31.08.2019 16:30


Computers and Technology, 31.08.2019 16:30

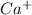 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 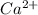 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 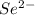 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 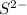 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)![Ca^+:19:[Ar]4s^1](/tpl/images/1191/0394/b80e3.png)
![Ca^{2+}:18:[Ar]](/tpl/images/1191/0394/ad5ef.png)
![Se^{2-}:32:[Ar]3d^{10}4s^24p^2](/tpl/images/1191/0394/c1b03.png)
![S^{2-}:18:[Ar]](/tpl/images/1191/0394/b3fdb.png)
![Ca:20:[Ar]4s^2](/tpl/images/1191/0394/46427.png)
![Se:34:[Ar]3d^{10}4s^24p^4](/tpl/images/1191/0394/0cf30.png)
![Se^{2-}:36:[Ar]3d^{10}4s^24p^6](/tpl/images/1191/0394/999f5.png) or
or ![Se^{2-}:36:[Kr]](/tpl/images/1191/0394/523d5.png)
![S:16:[Ne]3s^23p^4](/tpl/images/1191/0394/70407.png)
![S^{2-}:18:[Ne]3s^23p^6](/tpl/images/1191/0394/4203d.png) or
or 

