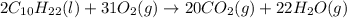Chemistry, 12.03.2021 23:20 cookies1164
Given below is the balanced equation for the combustion of decane. 2C10H22 (l) + 31O2 (g) --> 20CO2 (g) + 22H2O (g) What is the ratio of carbon dioxide molecules to oxygen molecules?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Given below is the balanced equation for the combustion of decane. 2C10H22 (l) + 31O2 (g) --> 20C...
Questions

English, 23.09.2020 08:01




History, 23.09.2020 08:01

History, 23.09.2020 08:01

Mathematics, 23.09.2020 08:01


Mathematics, 23.09.2020 08:01

Chemistry, 23.09.2020 08:01

History, 23.09.2020 08:01

Biology, 23.09.2020 08:01

Arts, 23.09.2020 08:01

Engineering, 23.09.2020 08:01

Mathematics, 23.09.2020 08:01

Health, 23.09.2020 08:01


Mathematics, 23.09.2020 08:01

Mathematics, 23.09.2020 08:01