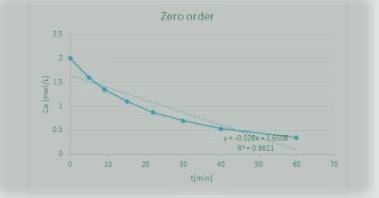
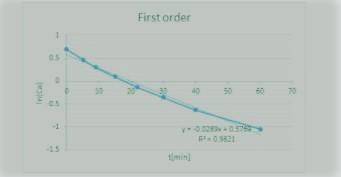
The Reaction
A+ B → C
was carried out in a constant-volume batch reactor where the following concentration measurements were recorded as a function of time:
t min 0 5 9 15 22 30 40 60
CA/mol/L 2 1.6 1.35 1.1 0.87 0.7 0.53 0.35
Required:
Determine the reactor order and the specific rate constant If you were to take more data, where would you place the points

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 13:00
In a copper wire, a temperature increase is the result of which of the following
Answers: 1

Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1
You know the right answer?
The Reaction
A+ B → C
was carried out in a constant-volume batch reactor where the foll...
was carried out in a constant-volume batch reactor where the foll...
Questions

English, 20.08.2019 05:00

Mathematics, 20.08.2019 05:00





Mathematics, 20.08.2019 05:10


Computers and Technology, 20.08.2019 05:10











![\mathbf{[A] = [A_o] -kt ---- (1)}](/tpl/images/1216/9615/f9d14.png)
![\mathbf{In([A])= In ([A_o])-kt ---- (2)}](/tpl/images/1216/9615/6a8a1.png)
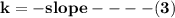
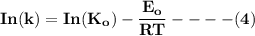
![\mathbf{In([A]) = In ([A_o])- kt}](/tpl/images/1216/9615/2a476.png)