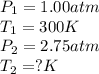Aflask of fixed volume contains 1.00 mole of gaseous carbon dioxide and 88.0 g of solid carbon dioxide. the original pressure and temperature in the flask is 1.00 atm and 300. k. all of the solid carbon dioxide sublimes. the final pressure in the flask is 2.75 atm. what is the final temperature in kelvins? assume the solid carbon dioxide takes up negligible volume.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 23.06.2019 01:00
Heat energy, carbon dioxide, and water are released through which process? a. photosynthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
Aflask of fixed volume contains 1.00 mole of gaseous carbon dioxide and 88.0 g of solid carbon dioxi...
Questions

Computers and Technology, 15.04.2020 20:56



Computers and Technology, 15.04.2020 20:56







Geography, 15.04.2020 20:56








Geography, 15.04.2020 20:57

Geography, 15.04.2020 20:57

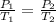 (at constant volume)
(at constant volume)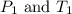 are the initial pressure and temperature of the gas.
are the initial pressure and temperature of the gas.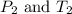 are the final pressure and temperature of the gas.
are the final pressure and temperature of the gas.