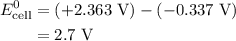Chemistry, 17.09.2019 07:30 nuggetslices
Calculate the standard emf of a cell that uses the mg/mg2+ and cu/cu2+ half-cell reactions at 25 °c. write the equation for the cell reaction that occurs under standard-state conditions and write the line notation for the cell.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1
You know the right answer?
Calculate the standard emf of a cell that uses the mg/mg2+ and cu/cu2+ half-cell reactions at 25 °c....
Questions

Mathematics, 27.09.2021 23:20


Mathematics, 27.09.2021 23:20


Biology, 27.09.2021 23:20

Social Studies, 27.09.2021 23:20




Advanced Placement (AP), 27.09.2021 23:20


History, 27.09.2021 23:20

English, 27.09.2021 23:20

Business, 27.09.2021 23:20


Spanish, 27.09.2021 23:20




English, 27.09.2021 23:20


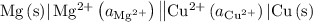
 .
.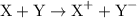
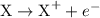

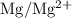 is
is 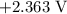 .
.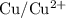 is
is 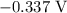 .
.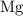 has higher oxidation potential thus the oxidation of
has higher oxidation potential thus the oxidation of 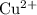 takes place at cathode.
takes place at cathode.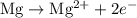 ......(1)
......(1)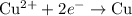 ......(2)
......(2)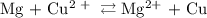 ......(3)
......(3)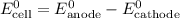 ......(5)
......(5)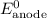 and
and 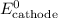 in equation (5).
in equation (5).