
Chemistry, 16.06.2021 07:30 babygirl123468
An inflated balloon contains X air molecules. After some time the volume of the balloon is found to be the half at the same temperature and pressure when a few air molecules are expelled out. a)How many molecules will be there in the balloon now? b) Which is the gas law associated with this?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

You know the right answer?
An inflated balloon contains X air molecules. After some time the volume of the balloon is found to...
Questions

Physics, 11.07.2019 15:30

Social Studies, 11.07.2019 15:30

Social Studies, 11.07.2019 15:30

Chemistry, 11.07.2019 15:30




Social Studies, 11.07.2019 15:30

Social Studies, 11.07.2019 15:30

Social Studies, 11.07.2019 15:30




English, 11.07.2019 15:30


English, 11.07.2019 15:30



English, 11.07.2019 15:30

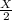 number of molecules now.
number of molecules now.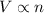 (At constant pressure and temperature)
(At constant pressure and temperature)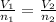 .....(1)
.....(1)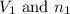 = Initial volume and number of moles
= Initial volume and number of moles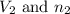 = Final volume and number of moles
= Final volume and number of moles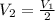
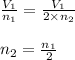
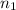 number of moles of gas contains X number of molecules
number of moles of gas contains X number of molecules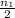 number of moles of gas will contain =
number of moles of gas will contain = 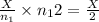 number of molecules
number of molecules

