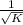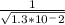At a given temperature, K = 1.3x10^-2 for the reaction:
N2(g) + 3H2(g) ⇌ 2NH3(g)
Calculate values of K for the following reactions at this temperature.
a. 1/2N2 + 3/2H2(g) ⇌ NH3(g)
b. 2NH3(g) ⇌ N2(g) + 3H2(g)
c. NH3(g) ⇌ 1/2 N2(g) + 3/2H2(g)
d. 2N2(g) + 6H2(g) ⇌ 4NH3(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2
You know the right answer?
At a given temperature, K = 1.3x10^-2 for the reaction:
N2(g) + 3H2(g) ⇌ 2NH3(g)
Calcul...
Calcul...
Questions

Computers and Technology, 05.02.2020 13:00

Mathematics, 05.02.2020 13:00

Mathematics, 05.02.2020 13:00


Biology, 05.02.2020 13:00



History, 05.02.2020 13:00



Mathematics, 05.02.2020 13:01




Computers and Technology, 05.02.2020 13:01



Health, 05.02.2020 13:01


English, 05.02.2020 13:01

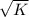
 = 0.11
= 0.11