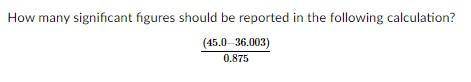How many significant figures in (45.0-36.003)/0.875 ?
...

Answers: 3


Another question on Chemistry



Chemistry, 23.06.2019 06:50
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1

Chemistry, 23.06.2019 10:30
Identify the limiting reactant when 9.65-g h2so4 reacts with 6.10-g of naoh.the equation is h2s04 + 2naoh = 2h2o + na2so4• what is the theoretical yield of na2so4, in grams? • how much of the excess reagent will remain after the reaction has been completed? • if 10.5-g of na2so4 are actually recovered experimentally, what is the percent yield?
Answers: 3
You know the right answer?
Questions



English, 07.12.2019 00:31

Social Studies, 07.12.2019 00:31



Mathematics, 07.12.2019 00:31



Mathematics, 07.12.2019 00:31