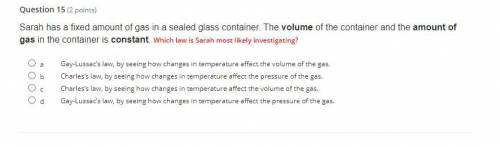
WILL MARK AS BRAINLIEST IF CORRECT
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

You know the right answer?
Questions


English, 08.11.2019 23:31



History, 08.11.2019 23:31

Mathematics, 08.11.2019 23:31


History, 08.11.2019 23:31


Mathematics, 08.11.2019 23:31

Chemistry, 08.11.2019 23:31


Mathematics, 08.11.2019 23:31

Health, 08.11.2019 23:31

History, 08.11.2019 23:31


Arts, 08.11.2019 23:31