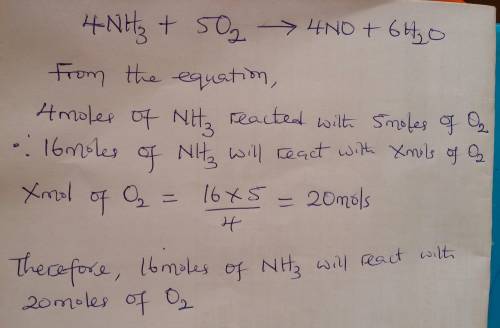Given the balanced equation representing a reaction:
4nh3 + 5o2 ==> 4no + 6h2o
what...

Chemistry, 17.11.2019 07:31 michelle5642b
Given the balanced equation representing a reaction:
4nh3 + 5o2 ==> 4no + 6h2o
what is the minimum number of moles of o2 that are needed to completely react with 16 moles of nh3?
(1) 16 mol (3) 64 mol
(2) 20. mol (4) 80. mol

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 21.06.2019 22:00
Which function is performed by earths atmosphere? a. ultraviolet rays are prevented from reaching the ozone layer. b. earths temperature is raised and moderated by trapping in heat c. charged particles from the sun are prevented from reaching earth. d. magnetic charges from space are prevented from reaching earths surface.
Answers: 2

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 13:30
Astudent is trying to create a table that compares hypotheses, theories, and laws. hypothesis theory law do scientific researchers formulate it? yes yes yes does it explain why things happen? yes yes no yes yes yes is it used to make predictions? no yes yes which of the following questions would most likely fill the blank in the table? is it an intelligent guess? is it newly formulated? is it based on observations? has it been proved?
Answers: 1
You know the right answer?
Questions

Mathematics, 07.06.2021 06:50



English, 07.06.2021 06:50


Biology, 07.06.2021 06:50


Law, 07.06.2021 06:50

Medicine, 07.06.2021 06:50

Mathematics, 07.06.2021 06:50


Mathematics, 07.06.2021 06:50

World Languages, 07.06.2021 06:50

Social Studies, 07.06.2021 06:50

Mathematics, 07.06.2021 06:50




Mathematics, 07.06.2021 06:50

Mathematics, 07.06.2021 06:50