Chemistry, 30.07.2019 11:30 nicolebastidas
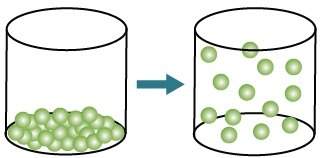
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a gas. atoms gain energy as a liquid changes to a gas. atoms lose energy as a solid changes to a gas. atoms lose energy as a liquid changes to a gas.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2

Chemistry, 23.06.2019 01:30
Which statement justifies that hydrogen peroxide (h2o2) is a polar molecule? the o – h bond is nonpolar and the molecule is asymmetric. the o – h bond is nonpolar and the molecule is symmetric. the o – h bond is polar and the molecule is asymmetric. the o – h bond is polar and the molecule is symmetric.
Answers: 1
You know the right answer?
How is energy related to the change of state represented by the model? atoms gain energy as a solid...
Questions

Mathematics, 09.09.2020 18:01


English, 09.09.2020 18:01


Mathematics, 09.09.2020 18:01

Mathematics, 09.09.2020 18:01


Social Studies, 09.09.2020 18:01



Mathematics, 09.09.2020 18:01

English, 09.09.2020 18:01


Physics, 09.09.2020 18:01


Mathematics, 09.09.2020 18:01

Mathematics, 09.09.2020 18:01



Advanced Placement (AP), 09.09.2020 18:01