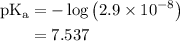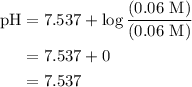Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
One mole of zinc has a mass of 65.4 grams. approximately how many atoms of zinc are present in one mole of zinc? 32 × 1023 atoms 6 × 1023 atoms 66 atoms 65 atoms
Answers: 1

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1
You know the right answer?
Calculate the ph of a solution formed by mixing 200.0 ml of 0.30 m hclo with 300.0 ml of 0.20 m kclo...
Questions


Mathematics, 01.02.2021 21:00


SAT, 01.02.2021 21:00

Mathematics, 01.02.2021 21:00

Chemistry, 01.02.2021 21:00





Social Studies, 01.02.2021 21:00

Computers and Technology, 01.02.2021 21:00


Mathematics, 01.02.2021 21:00

Mathematics, 01.02.2021 21:00



Physics, 01.02.2021 21:00

Mathematics, 01.02.2021 21:00

Computers and Technology, 01.02.2021 21:00

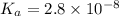
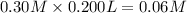
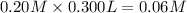
![pH=pK_{a}^o+\log\frac{[salt]}{[acid]}](/tpl/images/0135/8489/c0da1.png)
![pH=-\log[2.8\times 10^{-8}]+\frac{[0.06 M]}{[0.06 M]}](/tpl/images/0135/8489/ffe7d.png)
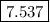 .
.
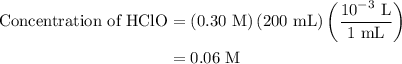
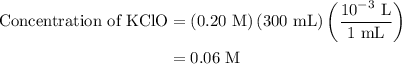
![{\text{pH}} ={\text{p}}{{\text{K}}_{\text{a}}} +\log \dfrac{{\left[ {{\text{KClO}}} \right]}}{{\left[ {{\text{HClO}}} \right]}}](/tpl/images/0135/8489/a099b.png) …… (1)
…… (1)
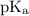 is the negative logarithm of acid dissociation constant.
is the negative logarithm of acid dissociation constant.
![\left[ {{\text{KClO}}} \right]](/tpl/images/0135/8489/79e91.png) is the concentration of salt, KClO.
is the concentration of salt, KClO.
![\left[ {{\text{HClO}}} \right]](/tpl/images/0135/8489/218b6.png) is the concentration of acid, HClO.
is the concentration of acid, HClO.
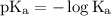 …… (2)
…… (2) 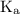 is the dissociation constant of HClO.
is the dissociation constant of HClO.
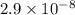 .
.