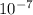Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 05:00
How many atomic mass units are equal to 1.672×10−24 g of protons?
Answers: 3

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
You know the right answer?
Boric acid frequently is used as an eyewash to treat eye infections. the ph of a 0.050m solution of...
Questions

Mathematics, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00


English, 09.12.2020 01:00


Mathematics, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00



Mathematics, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00




Physics, 09.12.2020 01:00



Mathematics, 09.12.2020 01:00

![\frac{[Acid]}{\text{[conjugate base]}}](/tpl/images/0115/9358/babe7.png)