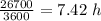Arsenic diffusion in si: arsenic is diffused into a thick slice of silicon with no previous arsenic in it at 1100ºc. if the surface concentration of the arsenic is 5.0 × 1018 atoms/cm3 and its concentration at 1.2 µm below the silicon surface is 1.5 × 1016 atoms/cm3 , how long must be the diffusion time? (d = 3.0 × 10-14 cm2 /s for as diffusing in si at 1100ºc.)

Answers: 1


Another question on Physics



Physics, 22.06.2019 18:30
4. now look at the green lines you created by connecting the three boiling point data points and the three melting point data points. for each of these lines, describe any trends you see. 5. locate the elements on your periodic table that you circled in green on your graph. what term or description would you use to identify these elements with respect to the periodic table? 7. using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers.
Answers: 2

Physics, 22.06.2019 20:00
Anika asks eva to roll a basketball and then a bowling ball to her. which requires more force to roll, and why?
Answers: 3
You know the right answer?
Arsenic diffusion in si: arsenic is diffused into a thick slice of silicon with no previous arsenic...
Questions

Mathematics, 07.10.2020 23:01










History, 07.10.2020 23:01




Engineering, 07.10.2020 23:01





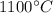
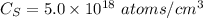
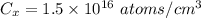
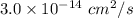
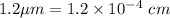
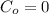
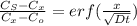
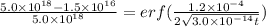
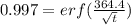 (1)
(1)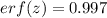
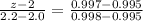
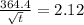
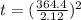 = 26700 s
= 26700 s