
Chemistry, 04.10.2019 22:00 leslieperez67
Tungsten is the metal used in the filaments of incandescent light bulbs. naturally occurring tungsten consists of five isotopes with the following isotopic masses and abundances:
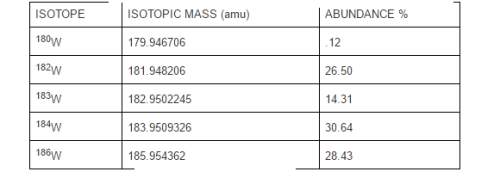

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Long term exposure to waves can cause sunburns and skin cancer. a) visible b) infrared c) gamma rays d) ultraviole
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 22:00
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3
You know the right answer?
Tungsten is the metal used in the filaments of incandescent light bulbs. naturally occurring tungste...
Questions

Social Studies, 07.10.2019 07:00


Biology, 07.10.2019 07:00

History, 07.10.2019 07:00


Mathematics, 07.10.2019 07:00



English, 07.10.2019 07:00


Mathematics, 07.10.2019 07:00




History, 07.10.2019 07:00


Mathematics, 07.10.2019 07:00

History, 07.10.2019 07:00


Social Studies, 07.10.2019 07:00



