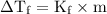Chemistry, 13.01.2021 07:20 thelordoftheknowwjo4
Which of the following aqueous solutions would have the greatest freezing-point depression?
(A) 0.10 m (NH₄)₂SO₄
(B) 0.10 m MnSO₄
(C) 0.10 m NaF
(D) 0.10 m KCI
(E) 0.10 m CH₃OH

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 18:40
What is one real world example of a colligative property?
Answers: 2

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2
You know the right answer?
Which of the following aqueous solutions would have the greatest freezing-point depression?
(A) 0.1...
Questions




Mathematics, 26.02.2020 19:55



Mathematics, 26.02.2020 19:56

Mathematics, 26.02.2020 19:56

History, 26.02.2020 19:56




Mathematics, 26.02.2020 19:56