
Chemistry, 15.07.2021 07:10 xonyemaa12
The equilibrium concentrations for the reaction between SO2 and O2 to form SO3 at a certain temperature are given in the table below. Determine the equilibrium constant and whether the reaction favors reactants, products, or neither at this temperature.
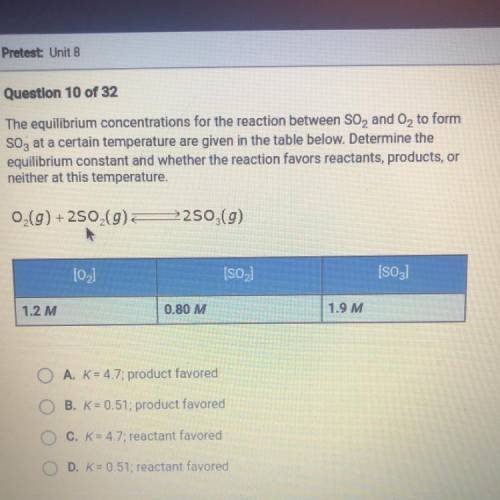

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
The equilibrium concentrations for the reaction between SO2 and O2 to form SO3 at a certain temperat...
Questions

Mathematics, 04.07.2019 12:30



History, 04.07.2019 12:30




Business, 04.07.2019 12:30

Mathematics, 04.07.2019 12:30

Chemistry, 04.07.2019 12:30



English, 04.07.2019 12:30


Mathematics, 04.07.2019 12:30


English, 04.07.2019 12:30

Mathematics, 04.07.2019 12:30


Mathematics, 04.07.2019 12:30



