
Chemistry, 12.11.2020 20:20 Britney8394
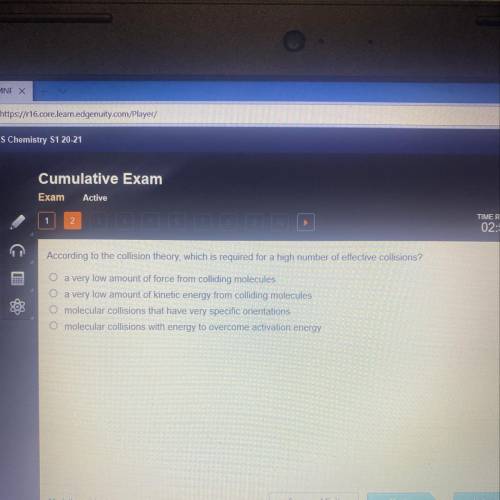
According to the collision theory, which is required for a high number of effective collisions?
a very low amount of force from colliding molecules
a very low amount of kinetic energy from colliding molecules
O molecular collisions that have very specific orientations
molecular collisions with energy to overcome activation energy

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

You know the right answer?
According to the collision theory, which is required for a high number of effective collisions?
a v...
Questions





Physics, 09.01.2020 05:31

















